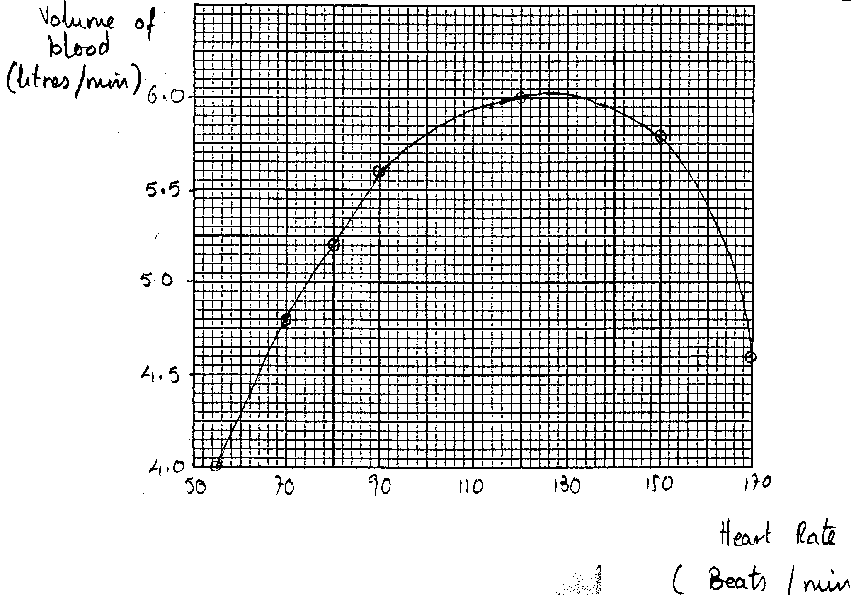This paper was published in the Journal of Education, 2012, Volume 7, Number 1, pp. 32-44.
(The title, 'My Language ∝ My Science) is inspired by Ludwig Wittgenstein’s ‘the limits of my language mean the limits of my world.’)
Abstract
This paper founded on my research extending over two
decades discusses the language demands of school science. These cannot be taken
for granted, especially when numerous initiatives are being undertaken to popularise
science subjects and thereby create a scientifically literate citizenry. The
paper accepts the position of many linguists and philosophers that language is
a key to understanding any subject and attempts to establish how ‘my world’ in
the Ludwig Wittgenstein’s famous quote, “the
limits of my language mean the limits of my world” (1922) also encompasses the
salient but often ignored “my science”.
Introduction
Each year examination reports at the CPE (Certificate of Primary
Education), SC (Cambridge School Certificate) and HSC (Cambridge Higher School
Certificate) levels indicate students’ inadequate language skills. Many Ordinary
‘O’ level and Advanced ‘A’ level science students are however not unhappy with
this weakness and openly flaunt their contempt for languages. They could not be
bothered with improving their language skills when they have the more important
job of studying the sciences. Their teachers ‘understand’ their difficulties
and the limited time they have to master the basic language skills and provide
support (which could be even in the form of essays on expected exam topics),
crack the examination code and dutifully ‘teach to the test’. Students cram the
expected and learn to the test for the examinations (Hunma, A., 2011; Hunma, V.,
2011) and manage to obtain ‘good’ results.
These practices are founded on the erroneous assumption that
science subjects being empirical in nature proceed through experimentation and
thus do not require the mastery of language skills. Beneath this is also some
false pride that languages are for not so bright kids, who study these because
they have no other options. Such attitudes and the subsequent teaching learning
practices further exacerbate the difficulties in science learning, especially for
Mauritians who learn it in a foreign language. To succeed, they therefore
require a robust mastery of both the highly specialised technical language of
science and the non-technical one for expression and communication.
Scientific literacy
One of the earlier attempts at defining the components of
scientific literacy was made by Pella and his colleagues in 1966. Many more
attempts have been made after this to define scientific literacy but the basic
elements have remained more or less the same and thus, it is the definition of
Pella et al., which is referred to in
this paper to discuss the ambit of science learning. They defined scientific
literacy as comprising of an understanding of the:
- the basic concepts of science;
- the nature of science;
- the ethics that the scientist in his/ her work;
- the interrelationships of science and society;
- the interrelationships of science and humanities;
- the differences between science and technology. (p.44)
There is a significant overlap among these
six components and it is not easy to address any one of them in isolation.
Nevertheless, this paper focuses mainly on the first component.
Understanding
of the basic concepts of science
Recently Yanish studying in Form I told me that he had learnt
about pasteurisation. To my questions;
“What is it?” “How is it done?” “Why is it done?” “What if it is not done?” his
answer, after some reflection was: “It improves the taste.”
The following week he was excited about an experiment with
purplish grey potassium permanganate crystals. He had dissolved the glistening
crystals in water and the water had turned uniformly purple. To my ‘when’,
‘why’ and ‘how’ questions, once again he had some vague ideas but clearly not
the underpinning science of diffusion of particles.
These are two very simple but new concepts that were introduced in
familiar contexts. Still, Yanish had difficulty explaining these. There are
many such students who are not able to explain their understanding of science. Could
this be because of their poor language skills?
Nevertheless, just before the exams, they will open their books
and learn the answers by heart and obtain good results and forget everything
soon afterwards.
The questions that we need to answer are as follows: Can we afford
to remain vague in our understanding of science? Do we need to learn it only
for the examination success? Will we still be successful in examinations if the
questions are set in unfamiliar contexts? How do we strengthen our science
learning?
It is useful to present another simple example here. At upper
primary and lower secondary levels, students are introduced to the states of
matter. The melting point of a solid is defined as the temperature at which a
solid changes to a liquid. The freezing point of a liquid is defined as the
temperature at which a liquid changes to a solid. Most students get these two
points separately. Nevertheless, it is not easy to appreciate the implication
that in theory and for most substances, the melting and freezing points are the
same temperature. Many years ago, a young student inferred this similarity and
got baffled. Both ice and water are at the same temperature! He tried to make
sense and shared his understanding with me. He explained that ‘for the ice the temperature was little below
the melting point’. How little? Why little?
At the
same time, introducing new concepts such as equilibrium, phase change /
transition, pressure, pure substance, nucleating substances or exceptions to
the rule, may further augment the difficulties. The questions that arise
concern what to teach and how to make young students understand.
Most Mauritian students have seen the Tamarin salt pans and know
that the water evaporates, leaving the salt crystals behind. Both crystallisation
and freezing are familiar processes. What are the subtle differences between
the two? How are the dissolving, melting, freezing, crystallisation and
evaporation, similar and different?
Students do try to make sense but science learning goes beyond
this personal construction of knowledge. Hodson (1988) points out:
Learning science is not simply a matter of
‘making sense of the world’ in whatever terms and for whatever reasons satisfy
the learner. Learning science involves introduction into the world of concepts,
ideas, understandings and theories that scientists have developed and
accumulated (that is, what science knows).
p.48
Driver et al. (1994) had also posited that “the process of knowledge construction must
go beyond personal empirical inquiry. Learners need to be given access not only
to physical experiences but also to the concepts and models of conventional
science.” p.7
This is a tall order. How does one achieve this progression from
personal constructions to knowledge constructions that have been socially
accepted by the scientific community? Language becomes important in this
transition as has been posited by many researchers such as Bruner (1964) and
Vygotsky (1978). They have established the importance of language in cognitive
development, understanding – how and why things are, and thinking about what
and how they could be.
The languages of science
As pointed
out in the preceding sections, science proceeds through two languages. The
first one is the highly specialized and technical language of science and the
other one is the non-technical one that is needed for expression and
communication. Students require the mastery of both to understand and become
active users of science. Consequently, it is crucial to ensure
mastery of the language in which science is taught, particularly in a
multilingual society where research studies by Tirvassen (2011) and
Raghoonundun (2011) have underlined the need for clearer language policy,
curriculum emphases and practices.
The technical language: It comprises the following:
- Familiar everyday words with new, specific and often different meanings,
- New words in familiar contexts,
- New words in new contexts,
- The semiotic or non-verbal language.
Moreover, some of these could be easily observable and
understandable at macro level and others could be abstract or at sub-micro
level requiring tacit understanding.
Familiar everyday words with specific meaning: Some terms, concepts and processes such as
mass, work, force, energy, current, power, pressure, time, salt, solution,
acid, base, element, compound, mole, boiling point, dissolving, diffusion,
root, fruit, egg, have definite meanings in science. For some, mole for
instance, the science meaning can be very different from its everyday
connotation.
Moreover, two terms with specific science may have only one word
in Kreol morisien. For example, both ‘dissolving’ and ‘melting’ are “fon” in Kreol morisien. Sugar dissolves
or ‘fon’ in water and ice melts or ‘fon’ to the liquid state.
New words in familiar context: The new words in familiar settings could include for
example, parts of a plant. The familiar “stem
is made of nodes and internodes, … Inside each root there are vessels to carry
water, mineral salts and food. A flower has sepals, petals, stamen and pistil.
The complete set of sepals is called the calyx. The complete set of petals is
called the corolla. A leaf has a main and lateral vein, each vein consists of
vessels to carry water, mineral salts and food….” (Science, Form 1,
Ministry of Education and Human Resources, 2006, pp. 28-30). Each new word has a specific meaning.
With the common experience of observing sugar and salt getting
‘moist’, students learn about deliquescence and hygroscopy and try to
understand the subtle difference. Deliquescence is the “action of absorbing water from air to form a solution” and hygroscopy
is the “action of absorbing water from air to become moist.” (Chemistry,
Form 3, Ministry of Education and Human Resources, 2005, p. 84)
New words in new contexts: The entirely new words in new settings could be cells, atoms,
sub-atomic particles, electronic structures, their configurations, …cathode,
anode, … vernier callipers, standard error, parallax error, potential energy, …
These also include technical terms, prefixes and suffixes in Latin
and Greek. For example, the animal invertebrate groups Cnidaria and
Echinoderms. “Cnidarians are aquatic
invertebrates. They have soft bodies with tentacles to catch small animals for
food. Hydra, sea anemone, polyps and jellyfish are examples of cnidarians.”
(Science, Form 1, Ministry of Education and Human Resources, 2006, p. 17).
“Echinoderms have a spiny
skin. Starfish, sea cucumber and sea urchin are examples of echinoderms.”
(ibid., p.19)
The above list of words constitutes only a small sample of the
vast scientific vocabulary that a student has to master.
There are many more at different levels of complexity that
students are required to learn, learn their spellings and right pronunciation
and understand the meaning that the scientific community has accepted. No
approximations are allowed.
The semiotic or non-verbal language:
Moreover, the technical language is not just verbal. There are
signs, symbols, numbers, abbreviations, images, equations, visual representations,
graphical representations. These cannot be used mechanically. One needs to
understand these for correct usage.
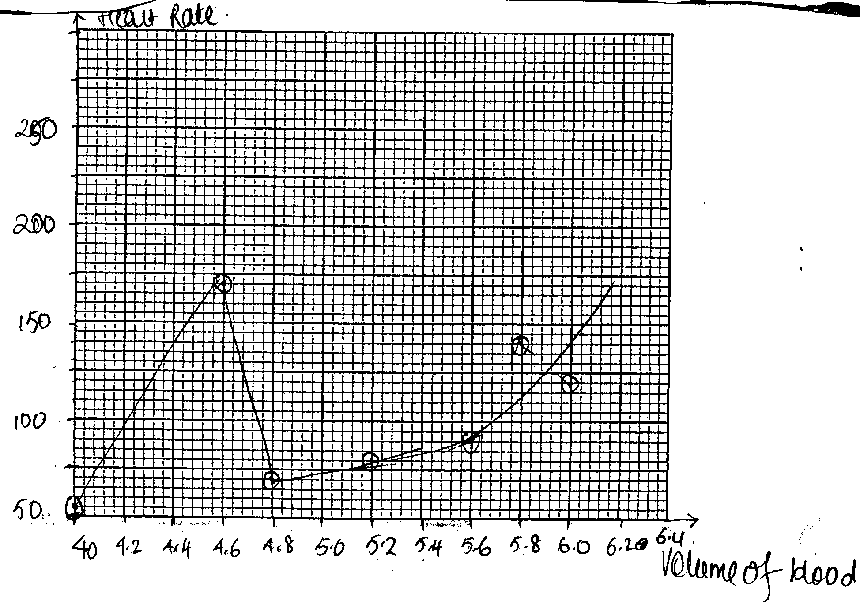
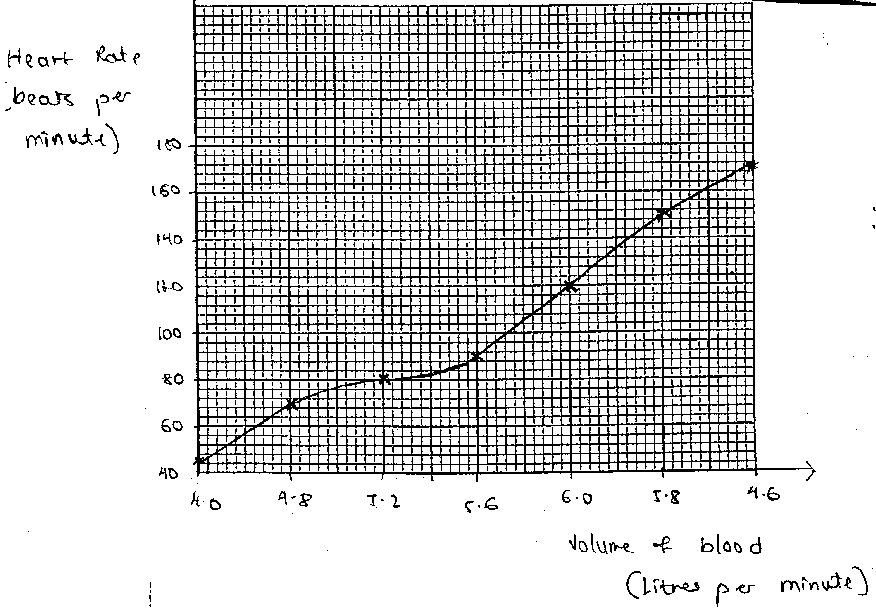
Graphical representation: As part of my PhD research (Hunma, 2003),
I asked a group of B.Sc. Year 1 students of the University of Mauritius to plot
a graph to present the data given below:
Use the data below to plot a graph:
Heart Rate: 55 70 80 90 120 150 170
(beats per minute)
Volume of blood: 4.0 4.8 5.2 5.6 6.0 5.8 4.6
(litres per minute)
This question was attempted by a total of
165 students in 1998 and 1999. Figures 1, 2, 3 and 4 represent the typical
graphs. Thirty-one students submitted plots similar to Figures 1 and 2. Six
students drew plots similar to Figure 3 and the remaining 128 drew the graphs
as shown in Figure 4. Figures 1, 2 and 3 raise many questions, especially when
graphs, their plotting and drawing inference form an important learning
outcome. About one fifth of the students could not plot the graph correctly.
Why did they not realise that there was something wrong with their graphs? Why
were the graphs plotted in such an unthinking way?
Figure 1
The non-technical language of science
Students come to the classroom with some scientific notions and
everyday experiences or what Claxton (1993) describes as ‘gut science’,
acquired through direct physical experiences and ‘lay science’ which is
acquired through social interaction and media experiences. Sometimes these
‘informal ideas’ (Black and Lucas, 1993) are acceptable. At other times they
need to be rejected or modified in line with the established scientific ideas.
Vygotsky (1962) calls these ideas ‘spontaneous concepts’. Students have
personal meanings for each one of these but these remain localised and
unrelated to other scientific concepts.
Teachers are expected to skilfully relate these spontaneous
concepts to the established knowledge and practices of science. This is not an
easy task because the learners may store the two – everyday science and the
formal science, acquired in two entirely different contexts in two separate
compartments with no apparent links between the two.
Moreover, this shift from the non-formal to the formal established
scientific knowledge becomes difficult because of students’ inadequate mastery
of the non-technical language. In fact, numerous research studies (See, for
example, Sharp, 1994) have established that science students have more
difficulties with the non-technical language than with technical language. In
an interesting study, Cassels and Johnstone (1985) came up with a list of
non-technical words that students had difficulty in using. Their list of
‘difficult’ words included words such as abundant, emit, linear, converse,
negligible and many others, the understanding of which we take for granted.
Students require the mastery of the non-technical language to understand and communicate the scientific knowledge in an objective and precise manner acceptable to the scientific community. As such, the language skills are required in order to:
- Listen to science lectures and talks,
- Read science texts,
- Talk about one’s science ideas,
- Write about science ideas.
Listen to science lectures and talks:
Most of the scientific knowledge is communicated to students through
teacher talk. Not all of it comprises the technical language of science.
Students need to develop the active listening skills that help
them in comprehending – what, when, why, how the new knowledge is related to
the much broader interdependent conceptual framework as well as retaining and
responding to the teacher talk.
Nevertheless, what often happens at the classroom is akin to
Wellington and Osborne’s (2001) ‘Postman Pat’ model of education. They state
that the “dominant metaphor for teaching
has now become ‘delivery’…. ‘Delivering learning’ as if it was some sort of
package or commodity which is passed on to the student, stored in a kind of
pigeon hole and later redelivered to a higher authority when assessment or
examination comes around.” (p.3)
In the absence of the active listening skills, many students
appear to be sincerely but thoughtlessly copying the teacher talk and then
mechanically memorising it for reproduction in examinations.
Read science texts: The reading of any written text involves scanning, skimming,
scrutinising for main ideas and the main train of arguments, inferring
meanings, understanding the relationships, separating facts from opinions,
drawing relevant conclusions and making informed judgments.
Science texts usually follow a strict procedure to present the
scientific knowledge. While reading a science text we learn about the knowledge
and also how it is to be communicated. The scientific community has a strict
regimen for both. This difficulty is further exacerbated because most reading
is done outside the class alone and without the teacher’s help.
Moreover, in general, most science texts are not easy to read.
This could be due to the nature of the subject where the authors are expected
to communicate the knowledge while adhering to
established conventions that include analytical and, impersonal style
with text written in third person.
Illustrations help but the difficulty level remains high.
One example is as follows: “The
locomotory system consists of the skeleton and muscles. The skeleton is made up
of bones. The skull, vertebral column (backbone) and the breastbone make up the
central part of the skeleton. The limb bones are connected to this part of the
skeleton and help in locomotion. The skeleton gives a form, shape and support
to the body parts. It also protects delicate organs such as heart, lungs and
the brain. Muscles are attached to the bones. They help to bring about
movements.” (Science, Form 1, Ministry of Education and Human Resources,
2006, p. 51)
This is a typical example of a science text which has nothing
superfluous. Each sentence introduces a new concept. The non-technical words
are essential, simple and specific – they illustrate the significant features
of the concepts.
Often with inadequate reading skills, students find science texts
difficult and resort to memorising the text without any understanding. In the
long run, they are not motivated to read more on their own.
At the same time, in most cases simplifying the texts is not the
solution because this can distort the picture or convey an incomplete one.
Talk about one’s science ideas: Talking about one’s science knowledge is an important
skill as it stimulates thinking and reflections on knowledge and learning.
Students discover inconsistencies in their thinking and reconstruct their ideas.
Teachers also come to know about students’ knowledge, its depth and
misconceptions.
Nevertheless, inside most science classes this student talk, as
Lemke (1990) describes it, is in the form of a ‘triadic’ dialogue where the teacher asks a question, student
replies and the teacher evaluates the answer.
There is hardly any discussion that could involve students’ views
and thereby give them an opportunity to test the robustness of their ideas.
(Hunma, V., 2011) There are many reasons for this absence of classroom
discussions. Students either lack the confidence to present their ideas or
hesitate to share their knowledge because of the severe competition that exists
among them. It could also be because of the excessive emphasis on course
coverage rather than on the quality of learning outcomes.
Write about science ideas: Students are expected to master the specific writing conventions
to communicate their knowledge of science.
Jones (2000) lists four different genres of science writing that
students should be aware of. These are “instruction,
explanation, argument and discussion.” (p. 100)
In addition, they need to write their reflections on their science
knowledge and their own learning to evaluate it and thereby promote it. Writing
thus engages their active participation, plays both the cognitive and
meta-cognitive roles and initiates the process whereby the students become
responsible for their own learning. However, for this, it is important that
students are given a lot of writing assignments that encourage them to
think about their science learning understanding rather than repeat what the
textbooks and their teachers state.
Conclusion
In this paper, an attempt is made to highlight some language
requirements of science and show how mastery of the language in which science
teaching and learning takes place is a necessary condition for understanding
scientific knowledge. This mastery promotes the understanding of – how and why
things are, and thinking about what and how they could be (Bruner, 1964;
Vygotsky, 1978). In a multilingual society, it becomes all the more
crucial to give the language science is taught in, its due importance in the
curriculum.
It is therefore for us to decide whether we want to encourage our students
to see the bigger picture of science and learn science for life rather than use
the easy and popular ‘delivery mode’ or the ‘Postman Pat’ model (Wellington, and Osborne, 2001)
of education for transient examination success.
References
Black, P. J. and Lucas, A.
M. (eds.), 1993, Children’s Informal
Ideas in Science. London: Routledge.
Bruner, J., 1964, The course of cognitive
growth, American Psychologist, 19, pp.1-16.
Cassels, J. and Johnstone,
A., 1985, Words that Matter in Science. London:
Royal Society of Chemistry.
Claxton, G.,1993,
Minitheories: a preliminary model for learning science. In P. J. Black and A.
M. Lucas (eds.) Children’s Informal Ideas
in Science. London: Routledge, p.45-61.
Driver, R., Squires, A., Rushworth, P. and
Wood-Robinson, V., 1994, Making Sense of
Secondary Science: Research into Children’s ideas, London: Routledge.
Hodson, D., 1998, Teaching and Learning Science: Towards a personalized approach,
Buckingham: Open University Press.
Hunma, A., 2011, Identities in transit and academic writing: an ethnographic study of first year Mauritian students at a South African university, Journal of Education, 6 (2), p.53-69.
Hunma, V., 2003, A Study of the Relationship between the Intended Curriculum and the Achieved Curriculum with Special Reference to Science Practical Work at HSC Level in Mauritius, Unpublished PhD Thesis, UoM.
Hunma, V., 2011, School Science and
Underachievement, Journal of Education,
6 (2), p.22-35
Jones, C., 2000, The Role of Language in
the learning and teaching of science, In M. Monk and J. Osborne (eds.) Good practice in science teaching, Buckingham,
Open University Press, p.88- 103.
Lemke, J. L., 1990, Talking Science: Language, Learning and Values, Norwood, NJ, Ablex.
Ministry of Education and Human
Resources., 2006, Science Form 1, EOI.
Ministry of Education and Human Resources.,
2005, Chemistry Form 3, EOI.
Pella, M.O., O’Hearn, G. T. and Gale, C. W., 1966, Scientific Literacy – Its Referents, The Science Teacher, 33 (5), p.44.
Raghoonundun, N., 2011., Clarté cognitive et littéracie : le développement de concepts scientifiques en langues étrangères en contexte d’enseignement multilingue, in R. Tirvassen (ed.) L’entrée dans le bilinguisme, l’Harmattan, pp.101-132.
Sharp, A., 1994., The
linguistic features of scientific English, School
Science Review, 75 (273) pp. 101-113.
Tirvassen, R., (2011) Curriculum et besoins langagiers en zone d’éducation
linguistics plurielle, Le Français dans
le monde. Recherche et application, 49, pp. 287-300.
Vygotsky, L.S., 1962, Thought and Language, Cambridge, MA, MIT Press.
Vygotsky, L.S., 1978, Mind in Society: The Development of Higher Psychological Processes,
Cambridge, MA, Harvard University Press.
Wellington, J. and Osborne,
J., 2001, Language and Literacy in
Science Education, Buckingham: Open University Press.
Wittgenstein, L., 1922, Tractatus Logico-Philosophicus, http://www.iep.utm.edu/wittgens/ accessed on 02.03.12.